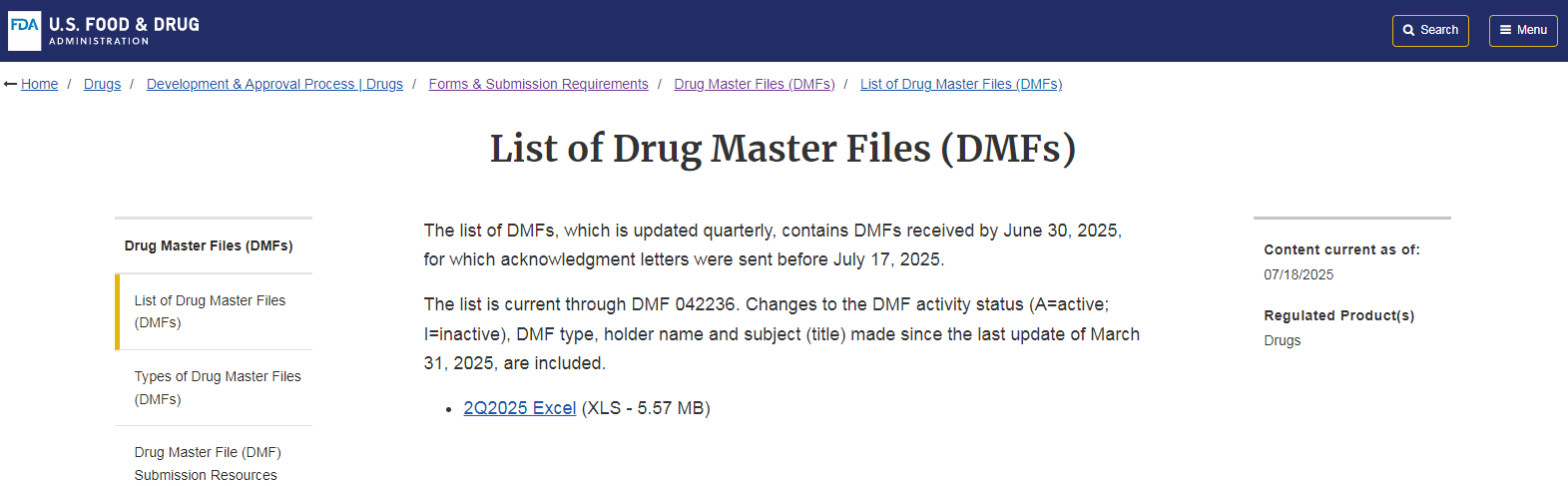
The U.S. Food and Drug Administration (FDA) website published that the tetrahydropyrimidine carboxylic acid (also known as ectoin) independently developed by Freda Biotechnology has successfully passed the DMF filing of the FDA for active pharmaceutical ingredients (API) (DMF filing No.038868), becoming one of the two products of the same type in the world that have passed the FDA API filing.
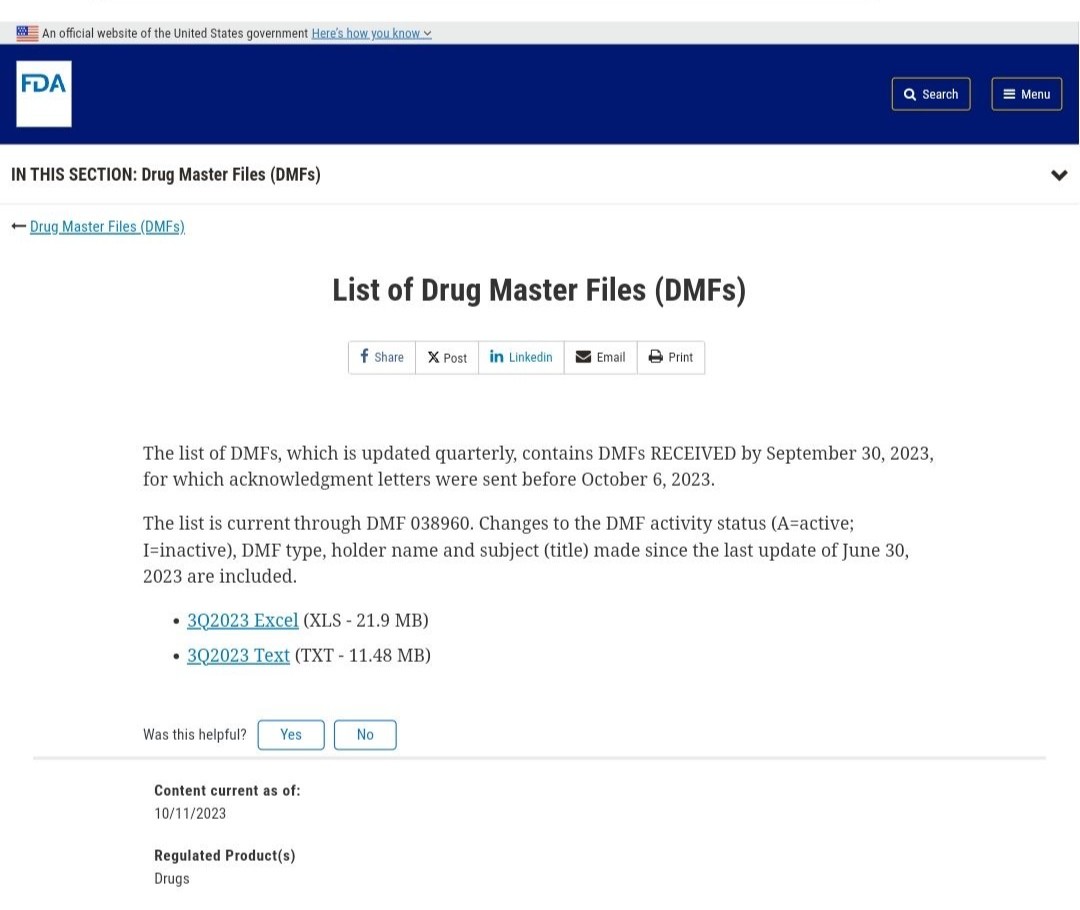
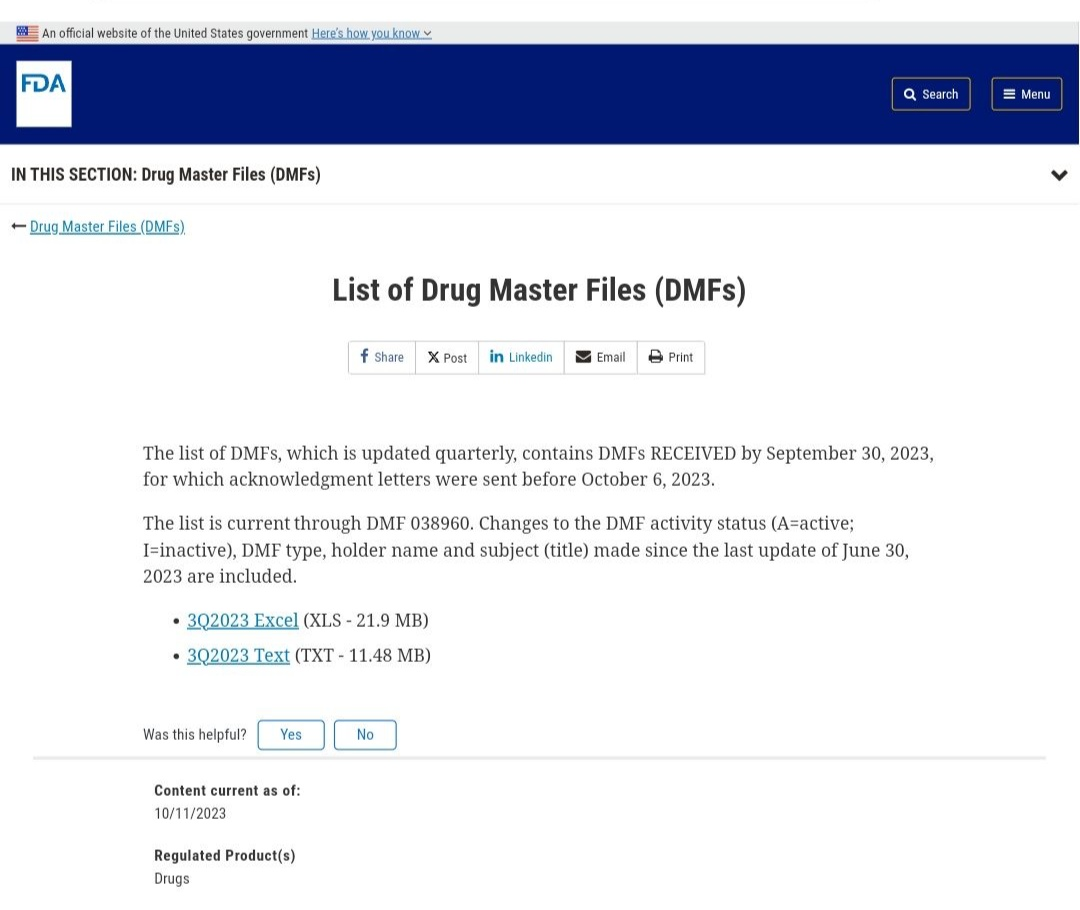
DMF stands for Drug Master File, which is an archival document submitted by a holder to the U.S. FDA. It contains confidential detailed information about the facilities, operational processes, raw materials, etc., used in the manufacturing, operation, packaging, and storage of drug products. Formulation manufacturers can directly reference the filing number, which significantly shortens the product review and evaluation time and accelerates the drug declaration process.
Next, the company will focus on the raw material sector planning of the group, leverage its technical advantages in the raw material segment, and simultaneously carry out basic and applied research on new raw materials to expand their application scope. It will further layout overseas markets to provide higher-quality services for more internationally renowned brands.
Related recommendations
PRODUCTS
About us
Leave a Message
Your email address will not be published. Required fields are marked *